For Food Processing Operations Managers, the landscape of regulatory compliance is undergoing its most significant transformation since the initial rollout of the Food Safety Modernization Act (FSMA). The introduction of FSMA Section 204(d)—commonly referred to as the Final Rule on Requirements for Additional Traceability Records for Certain Foods—establishes a new paradigm for record-keeping. No longer is “one step forward, one step back” sufficient for items on the Food Traceability List (FTL). Instead, the FDA now mandates a comprehensive end-to-end digital footprint designed to accelerate outbreak response and minimize the scope of product recalls.
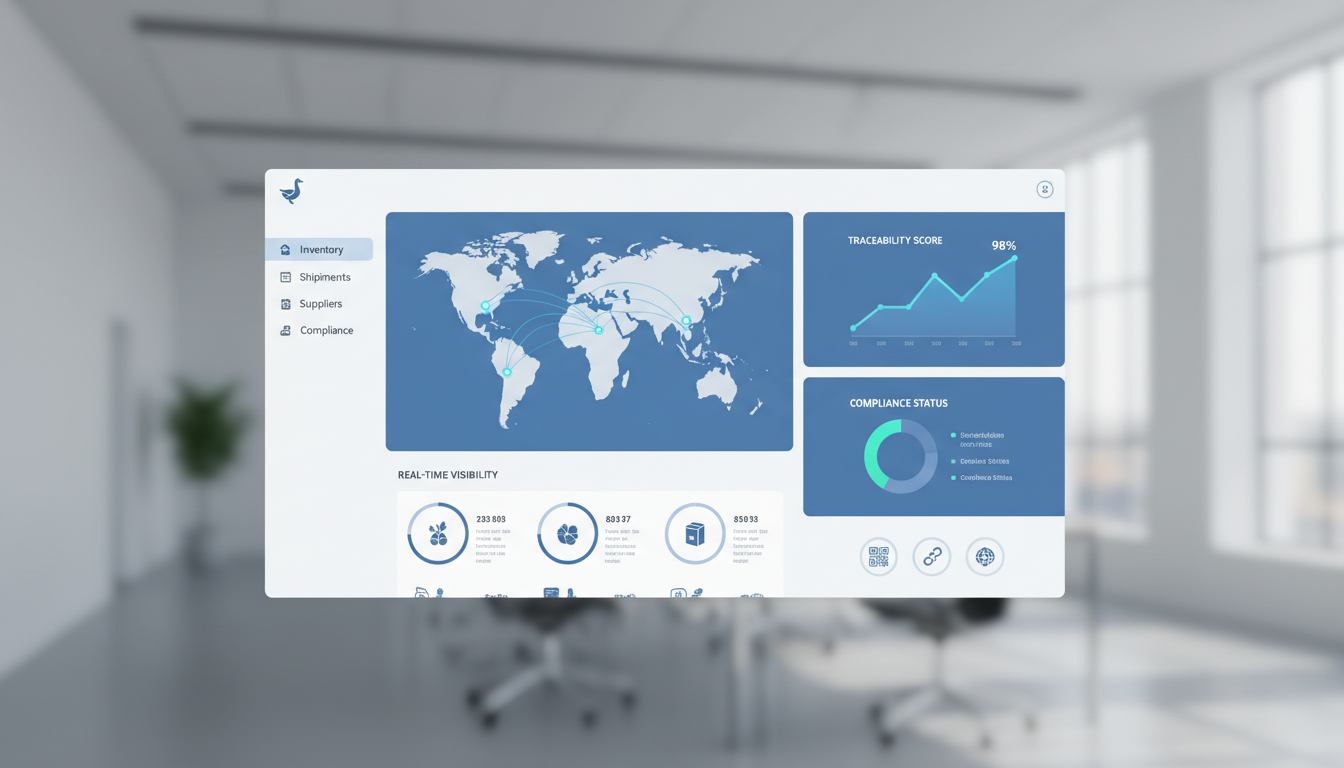
As the January 20, 2026, compliance deadline approaches, facilities must move beyond manual logs and fragmented spreadsheets. Central Valley Cold Storage (CVCS) has anticipated this shift by integrating the “Goose” system, a sophisticated, web-based inventory management platform designed to automate the heavy lifting of data acquisition. This FSMA 204 Guide examines the technical requirements of the new rule and how digital traceability systems act as the primary defense for modern logistics operations.
Understanding FSMA 204
The core objective of FSMA 204 is the standardization of data across the entire supply chain. Historically, food safety investigations were often delayed by incompatible record-keeping formats, illegible handwritten logs, and the time-consuming process of reconciling disparate lot codes. FSMA 204 solves this by requiring specific “Key Data Elements” (KDEs) to be recorded at “Critical Tracking Events” (CTEs).
Critical Tracking Events (CTEs)
For an Operations Manager, understanding CTEs is fundamental to workflow design. The FDA identifies several specific events that trigger record-keeping requirements:
- Harvesting: The initial point of origin for raw agricultural commodities.
- Cooling: The initial temperature management phase, critical for the cold chain integrity.
- Initial Packing: The first time a food item is placed into a container that will reach the consumer.
- First Receiver: The first person (other than a farm) who purchases and takes physical possession of a food on the FTL.
- Shipping: Any movement of food from one location to another.
- Receiving: The point at which a food is received by a processor or distributor.
- Transformation: Any process that changes a food (e.g., cutting, cooking, or commingling ingredients).
Key Data Elements (KDEs)
For every CTE, a specific set of KDEs must be captured. This includes the Traceability Lot Code (TLC), the location description of where the event occurred, the date and time, and the specific quantity of the product. The technical challenge lies in the “Traceability Lot Code” itself. Under FSMA 204, the TLC must be assigned at the point of initial packing or transformation and must remain linked to the product as it moves through the supply chain. Changing or reassining lot codes without proper cross-referencing is a primary source of non-compliance.
The Food Traceability List (FTL)
Not all food products fall under the stringent requirements of FSMA 204. The FDA has curated the Food Traceability List (FTL) based on a risk-ranking model that considers the frequency of outbreaks and the severity of illness associated with specific commodities. However, for most diversified processing facilities, the FTL covers a broad spectrum of inventory.
Supporting data from the FDA confirms that the FTL includes, but is not limited to:
- Fresh Produce: Leafy greens (including fresh-cut salads), melons, peppers, tomatoes, cucumbers, and herbs.
- Nut Butters: All varieties of nut and seed butters.
- Seafood: Finfish (including smoked fish), crustaceans, and molluscan shellfish.
- Dairy: Soft cheeses and deli salads.
- Eggs: Shell eggs from poultry.
For Operations Managers, the complexity arises when FTL items are used as ingredients in non-FTL products. For example, if fresh-cut peppers (an FTL item) are included in a pre-packaged fajita kit, the entire kit may become subject to traceability requirements during the “Transformation” CTE. This “cascading compliance” is why a robust digital system like Goose is no longer an optional luxury but a core operational necessity.
How the Goose System Automates KDEs
The technical burden of FSMA 204 is the requirement for 24-hour record retrieval. If the FDA requests traceability data, the facility must provide an electronic sortable spreadsheet within one business day. For facilities relying on paper, this is an almost impossible task. The Goose system, utilized by Central Valley Cold Storage, is engineered to handle this high-velocity data environment through automation.
Automated Inbound and Outbound Mapping
When a shipment arrives at our facility, the Goose system captures the Receiving CTE data instantaneously. By scanning GS1-128 barcodes or integrated RFID tags, the system populates the KDEs—including the Traceability Lot Code, the source location, and the entry time—directly into a secure, web-based portal. This eliminates manual entry errors and ensures that the digital twin of the product matches the physical inventory.
Digital Lot and Commodity Mapping
The Goose system excels at “Transformation” tracking. In food processing, commodities are often moved from bulk storage into smaller units or combined into blends. Goose creates a digital genealogy for every lot. If a processor uses three different lots of leafy greens to create a single batch of mixed salad, the Goose system maintains the link to all three parent TLCs. This granularity ensures that in the event of a recall, only the affected lots are pulled, rather than the entire day’s production.
The 24-Hour Retrieval Guarantee
Because Goose is a cloud-native platform, Operations Managers have 24/7 access to traceability logs. During a safety audit or an FDA inquiry, users can generate the required electronic sortable spreadsheets with a few clicks. This speed not only ensures compliance but also demonstrates a high level of professional competence to inspectors and clients alike, further solidifying the facility’s certifications and reputation.
| FSMA 204 Requirement | Goose System Solution |
|---|---|
| 24-Hour Record Retrieval | Instant Web Portal Access |
| CTE Tracking | Automated Inbound/Outbound Logs |
| KDE Documentation | Digital Lot & Commodity Mapping |
| Sustainability | Paperless Digital Auditing |
Liability Reduction for Processors
For an Operations Manager, FSMA 204 compliance is as much about risk management as it is about regulatory adherence. The financial cost of a recall is often measured in the millions, but the reputational damage can be permanent. Digital traceability acts as a “surgical tool” for recalls.
Minimizing Recall Scope
Without precise digital records, companies often have to issue “blanket recalls,” pulling all products produced within a broad timeframe. With the Goose system’s automated KDE tracking, CVCS allows growers and processors to identify the exact pallet, the exact row, and the exact time a specific lot was handled. This precision can reduce the volume of recalled product by up to 90%, saving massive amounts of capital and preventing unnecessary food waste.
Audit Readiness and Sustainability
The shift to paperless digital auditing is a significant component of modern sustainability goals. Beyond the environmental impact of reducing paper, digital systems create a permanent, immutable audit trail. In a manual system, records can be lost, damaged, or altered. The Goose system provides a time-stamped, encrypted log of all inventory movements. This high-integrity data reduces the liability of the Operations Manager by providing undeniable proof of due diligence and cold chain maintenance.
Enhanced Visibility for Growers
Central Valley Cold Storage offers automated reporting for growers, providing them with real-time insights into their inventory status. This transparency builds trust and allows for better production planning. When growers know that their logistics partner is utilizing a state-of-the-art “FSMA 204 Guide” compliant system, they are more likely to engage in long-term partnerships, knowing their product is safe and their compliance is handled.
Conclusion
The transition to FSMA 204 is a technical challenge that demands a technological solution. For Operations Managers, the stakes involve both public safety and operational continuity. By moving toward digital traceability now, facilities can avoid the chaos of the 2026 deadline and begin reaping the benefits of increased efficiency and reduced liability today.
Central Valley Cold Storage, powered by the Goose system, provides the infrastructure necessary to navigate these complex regulations. Our commitment to automated KDE capture, real-time visibility, and 24-hour record retrieval makes us the premier choice for processors handling FTL commodities. In the world of modern logistics, data is just as important as the temperature of the warehouse. Ensure your data is as cold and as controlled as your product.
Frequently Asked Questions
Q: What is the deadline for FSMA 204?
A: The compliance date is January 20, 2026, but the Goose system enables immediate adoption, allowing firms to troubleshoot their workflows and ensure data accuracy well before the regulatory enforcement begins.
Q: Does FSMA 204 apply to frozen foods?
A: It depends on the commodity. While many frozen items are excluded if they undergo a “kill step” or remain frozen, many fresh-frozen ingredients on the FTL still require stringent traceability records throughout the cold chain.
Don’t let the 2026 deadline catch you off guard. Experience the power of automated traceability and see how the Goose system can streamline your operations.